Home / Online GXP courses / GMP : Good Manufacturing Practices / Corrective Action and Preventive Action (CAPA) Training


Successful completion provides learners with a dated, traceable and downloadable certificate for the course :
Each certificate is CPD/CEU accredited and 21 CFR PART 11 compliant. Certificates validity can be checked online through our certificate online checker. They can also be shared on Linkedin.
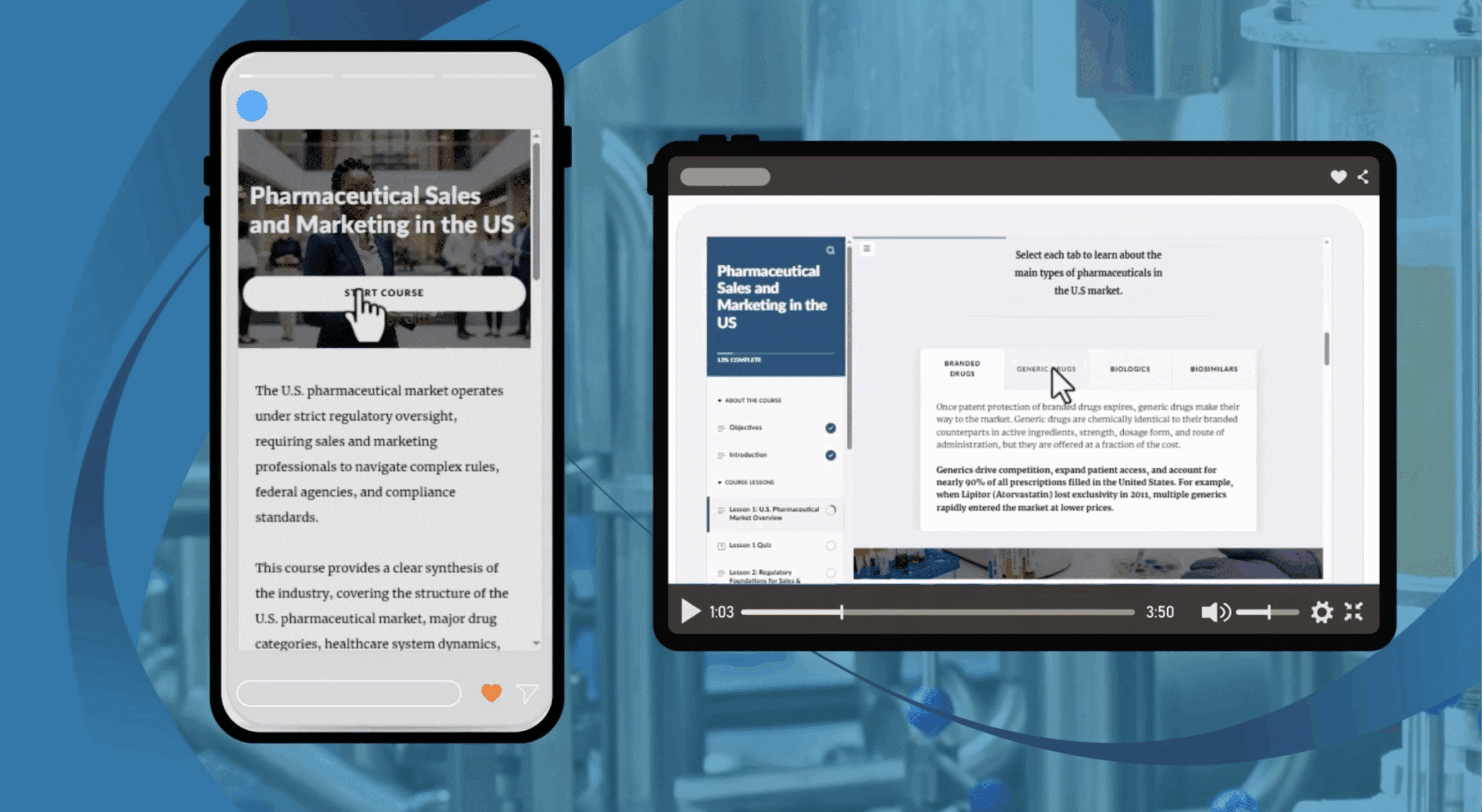
Corrective Action and Preventive Action (CAPA) is a core FDA GMP requirement used to investigate nonconformities, identify root causes, and prevent issues from recurring in pharmaceutical and medical‑device operations. This course provides a practical introduction to CAPA principles under FDA 21 CFR 820.100, using real‑world examples to show why effective CAPA is essential.
Upon successful completion, you will receive a validated certificate accredited with CPD/CEU credits, suitable for audit and compliance documentation.
Who should enroll?
What you’ll learn?
Introduction
Lesson 1: What is CAPA?
Lesson 2: Purpose of CAPA
Lesson 3: CAPA Requirements
Lesson 4: CAPA Procedures
Lesson 5: Objectives of CAPA
Lesson 6: Filling The CAPA Template Form
Glossary
Conclusion
Final assesment


Large group ? Request a quote !


$95.00 / year
$95.00 / year