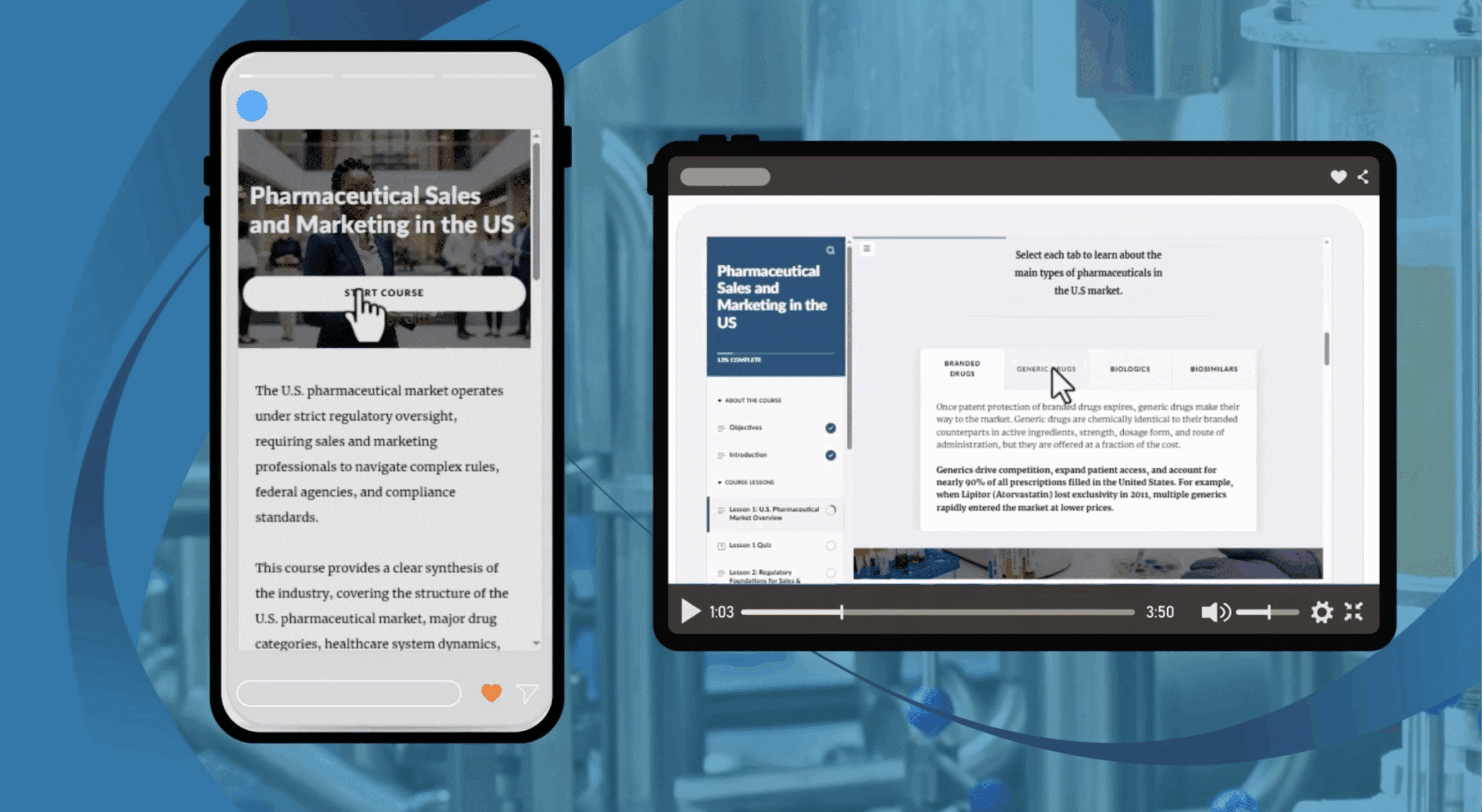
Home / Online GXP courses / Cross-Industry / Management of Service Providers


Successful completion provides learners with a dated, traceable and downloadable certificate for the course :
Each certificate is CPD/CEU accredited and 21 CFR PART 11 compliant. Certificates validity can be checked online through our certificate online checker. They can also be shared on Linkedin.
A fool-proof service provider recruitment program provides a barrier for only materials and services of acceptable quality in the manufacturing process.
This course shows the importance of following a uniform recruitment process. The process should include the evaluation of the service provider’s reputation in the market and the quality of the materials or services they provide.
A credible monitoring process ensures that the quality provided is consistent. Hence, your recruitment team should include qualified professionals. Consequently, the company’s image will be preserved and the consumers interests protected.
In this course, we provide a synthesis of the topic through an interactive online experience. This material was built by a team of Regulatory Affairs Experts with a Qualification from Northeastern University, Boston, piloted by a Senior Member able to articulate theory with practice.
Lesson 1: Who is a Service Provider?
Lesson 2: Identification
Lesson 3: Evaluation and Assessment
Lesson 4: Qualification
Lesson 5: Audits
Lesson 6: Agreements
Conclusion
Evaluation
Glossary


Large group ? Request a quote !


$95.00
$95.00