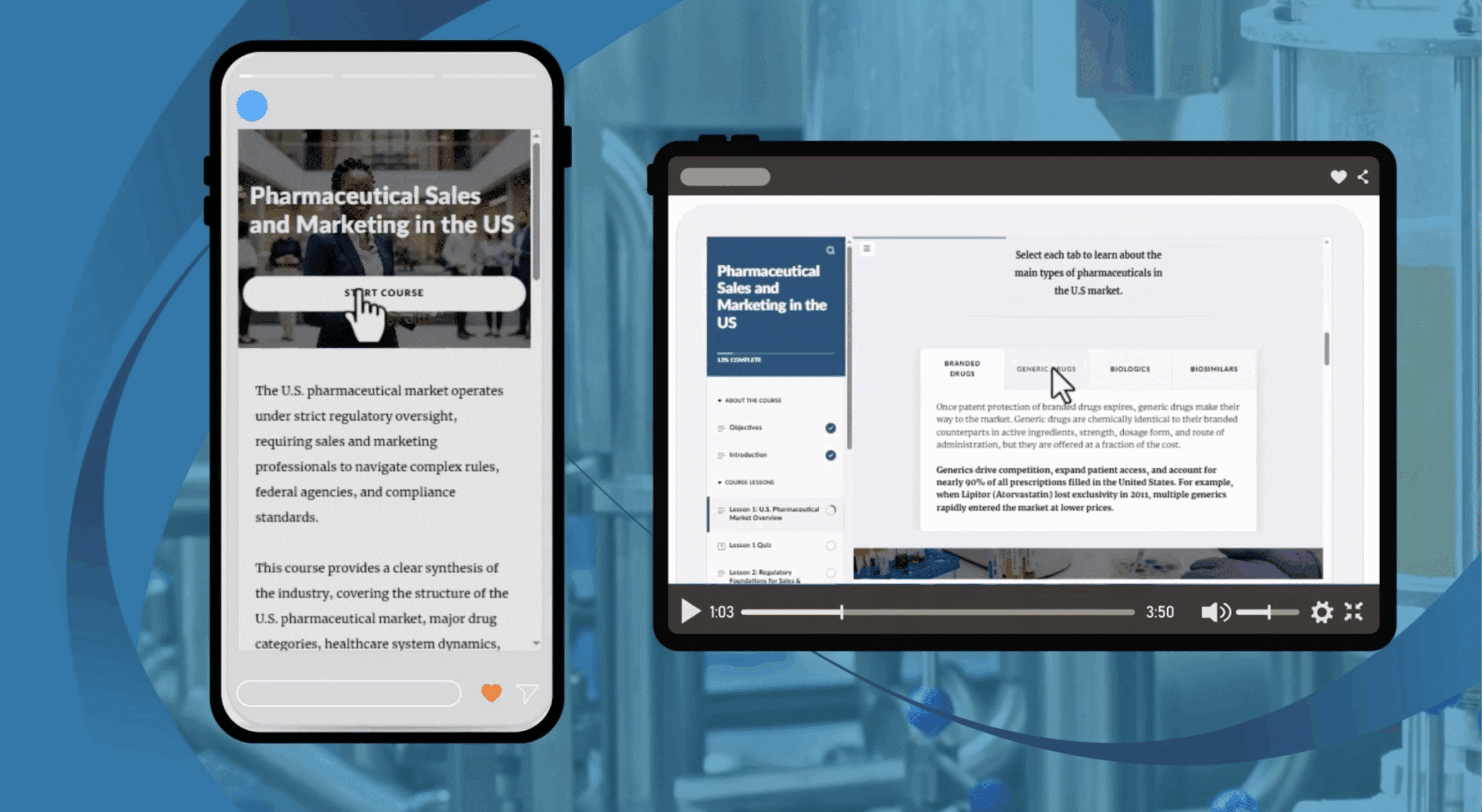
Home / Online GXP courses / Medical Devices / GMP for Medical Devices and FDA 21 CFR PART 820


Successful completion provides learners with a dated, traceable and downloadable certificate for the course :
Each certificate is CPD/CEU accredited and 21 CFR PART 11 compliant. Certificates validity can be checked online through our certificate online checker. They can also be shared on Linkedin.
All common medical procedures require medical devices and they are used by laypersons at home and by healthcare professionals in medical facilities. From palliative care to diagnosis, prevention, and screening, medical devices can do it all. It is therefore vital that they comply with the highest standards of quality.
GMP or Good Manufacturing Practice for medical devices refers to a set of guidelines and regulations which ensure that medical devices are consistently produced with high quality standards. Compliance with GMP for medical devices is important as it ensures the quality, safety, strength, and purity of the final product.
This online training also covers aspects such as storage, distribution, installation and servicing, and the provision of associated services
Introduction
Lesson 1: Design Controls of Medical Devices
Lesson 2: Document Controls of Medical Devices
Lesson 3: Purchasing Controls of Medical Devices
Lesson 4: Identification and Traceability
Lesson 5: Production and Process Controls
Lesson 6: Acceptance Activities
Lesson 7: Requirements for Nonconforming Product
Lesson 8: Corrective and Preventive Action
Lesson 9: Labeling and Packaging Controls of Medical Devices
Lesson 10: Handling, Storage, Distribution, and Installation of Medical Devices
Lesson 11: Records
Lesson 12: Servicing and Statistical Techniques
Glossary
Evaluation


Large group ? Request a quote !


$95.00 / year
$95.00 / year