Home / Online GXP courses / Pharmaceuticals / Pharmaceutical Sales and Marketing in the US


Successful completion provides learners with a dated, traceable and downloadable certificate for the course :
Each certificate is CPD/CEU accredited and 21 CFR PART 11 compliant. Certificates validity can be checked online through our certificate online checker. They can also be shared on Linkedin.
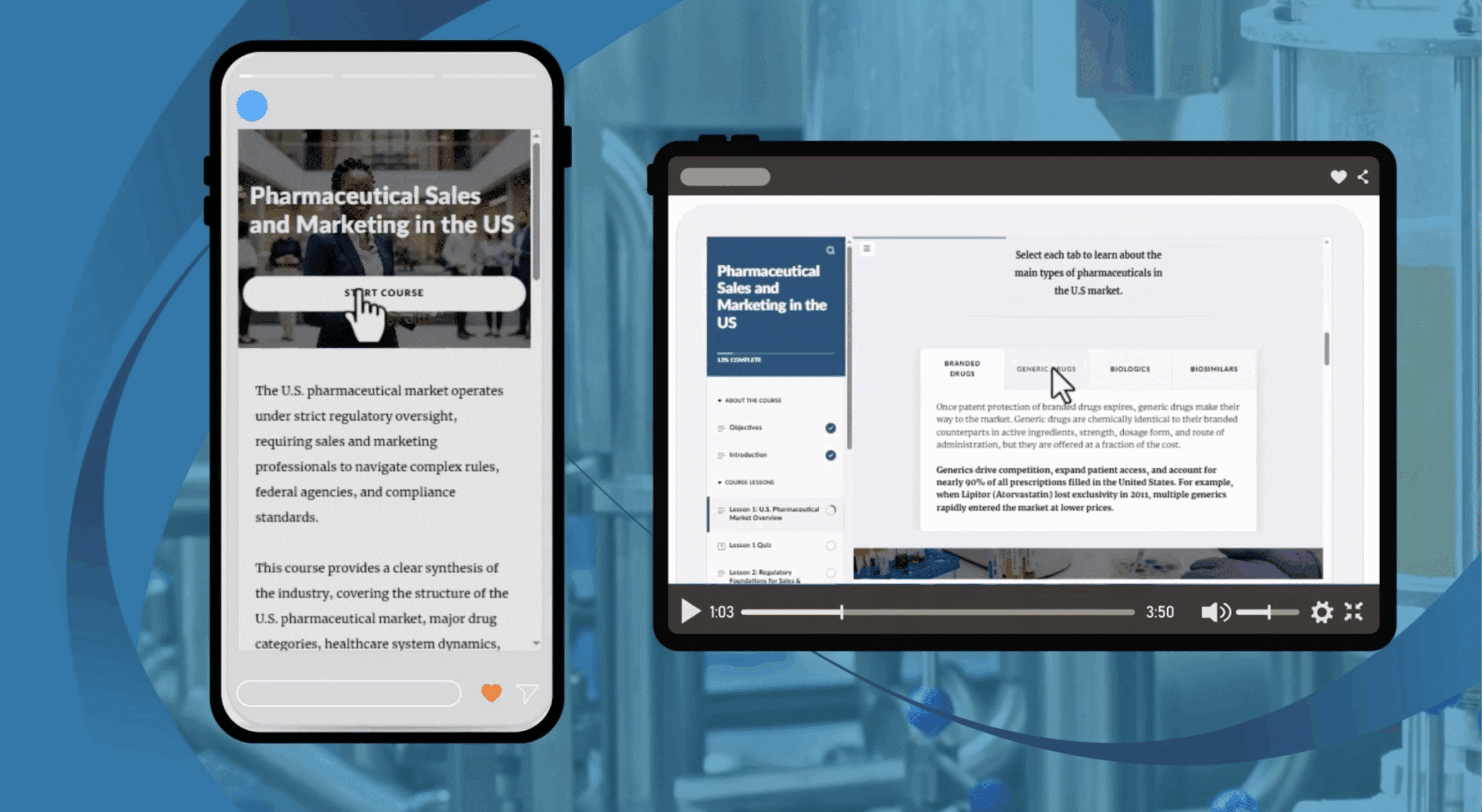
The U.S. pharmaceutical market is a rapidly evolving and highly regulated environment, where effective sales and marketing require a strong understanding of compliance, market structure, and patient access. As the industry continues to grow, professionals must navigate complex rules while driving ethical and sustainable commercial success.
This course provides a practical overview of how pharmaceutical sales and marketing operate within the U.S. healthcare ecosystem, covering market structure, drug categories, patient pathways, and the regulatory and compliance frameworks that govern promotional activities.
The content is developed by industry experts with direct experience in the U.S. pharmaceutical market, ensuring alignment with real-world regulatory, commercial, and ethical standards.
Upon successful completion, you will receive a validated certificate accredited with CPD/CEU credits, suitable for audit and compliance documentation.
✅ Who should enroll?
🎯 What you’ll learn?
Lesson 1: U.S. Pharmaceutical Market Overview
Lesson 2: Regulatory Foundations for Pharmaceutical Sales and Marketing
Lesson 3: Compliance Frameworks and Industry Standards
Lesson 4: Ethical Sales Practices and Essential Field Skills
Lesson 5: Market Access, Pricing, and Reimbursement
Lesson 6: Marketing Strategies and Digital Innovation in Pharma
Conclusion
Assessment


Large group ? Request a quote !


$95.00 / year
$95.00 / year