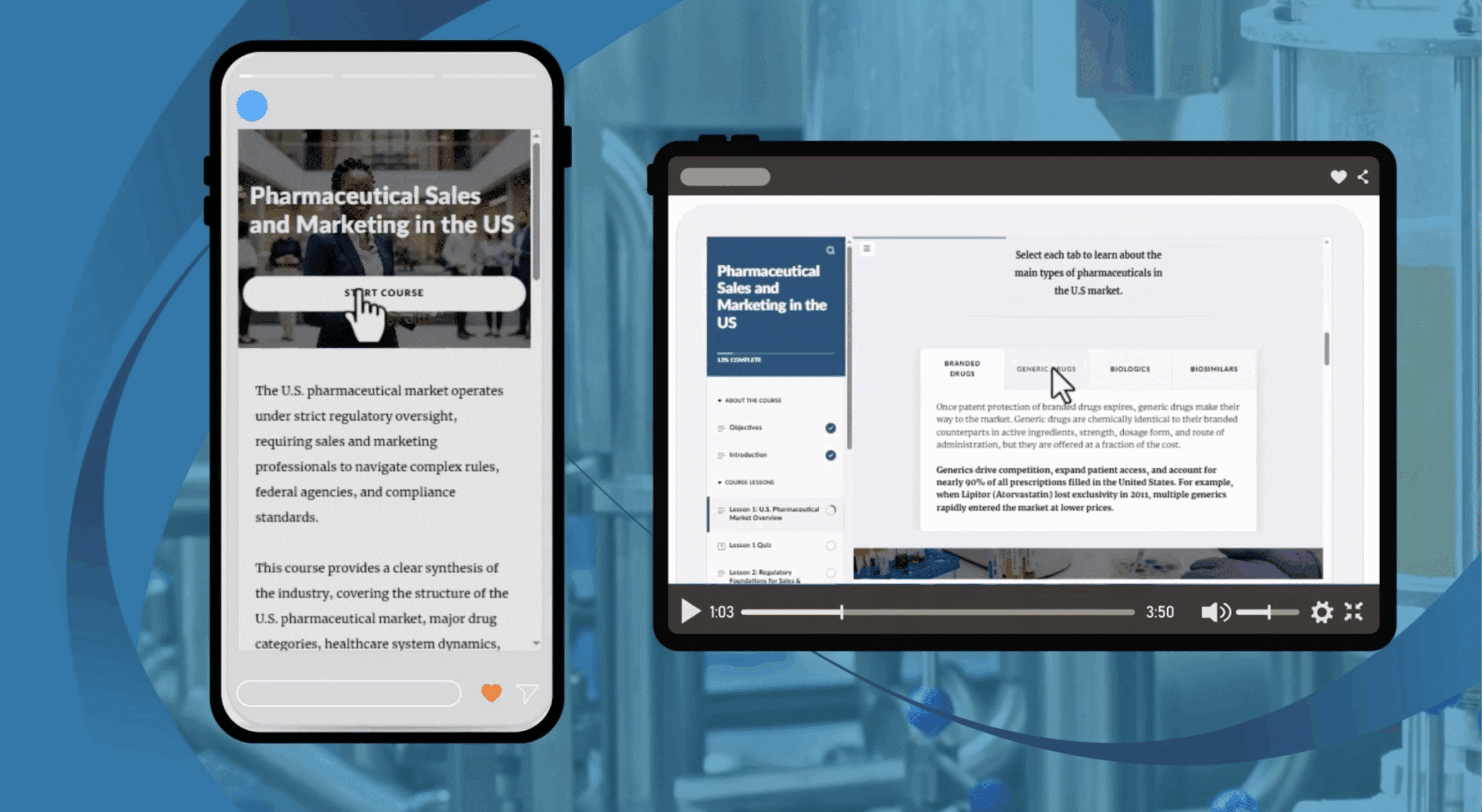
Home / Online GXP courses / GCP : Clinical Trials / General Data Protection Regulation (GDPR) for Pharmaceuticals and Clinical Trials


Successful completion provides learners with a dated, traceable and downloadable certificate for the course :
Each certificate is CPD/CEU accredited and 21 CFR PART 11 compliant. Certificates validity can be checked online through our certificate online checker. They can also be shared on Linkedin.
Life‑sciences organizations handle some of the most sensitive personal data in the world, making GDPR compliance essential for protecting patient privacy, maintaining trust, and avoiding costly regulatory penalties. Clinical trials, pharmacovigilance activities, and global research collaborations all require a clear understanding of how GDPR applies to health data.Developed by experts in data protection and clinical operations, the course also covers international data transfers, especially EU‑to‑US transfers, ensuring compliance throughout the clinical‑trial lifecycle.
🚀 Upon successful completion, participants receive a downloadable, internationally recognized GxP training certificate, accredited with CPD/CEU credits, suitable for professional and HR records.
✅ Who should enroll?
This course is perfect for data protection officers, compliance managers, and clinical research professionals managing patient data. It also benefits regulatory affairs specialists, quality assurance personnel, and pharmaceutical or biotech professionals handling sensitive data, ensuring GDPR compliance across life sciences organizations.
🎯 What you’ll learn?
Introductory Video
Lesson 1: Personal Data in Clinical Research
Lesson 2: Controllers in Clinical Data Protection
Lesson 3: Processors in Clinical Data Protection
Lesson 4: Consent
Lesson 5: Rights of Data Subjects
Lesson 6: Data Protection Impact Assessment (DPIA)
Lesson 7: Handling International Transfers of Clinical Data
Conclusion
Evaluation


$95.00 / year
$95.00 / year